 %20with%20water.jpg)
Your browser does not support the audio element. When moving down the periodic table, each alkali metal has an increasing atomic radius as well as increasing reactivity.The compound ammonium has very similar properties to the heavier alkali metals.They are considered to be more similar to each other than any other group in the periodic table. Reactivity depends on the loss of the outer electron, and so it increases down the group, because the outer electron is less firmly held as you go down the.All alkali metals have odd atomic numbers.Different alkali metals burn with different colored flames including sodium (orange yellow), lithium (red), potassium (lilac), rubidium (red), and cesium (blue or violet). Brief statement of the principal resemblances of elements within each main group, in particular alkali metals, alkaline earth metals, halogens and noble gases.The name "alkali" is derived from the Arabic word for "ashes.".They like to form salts by combining with halogens. 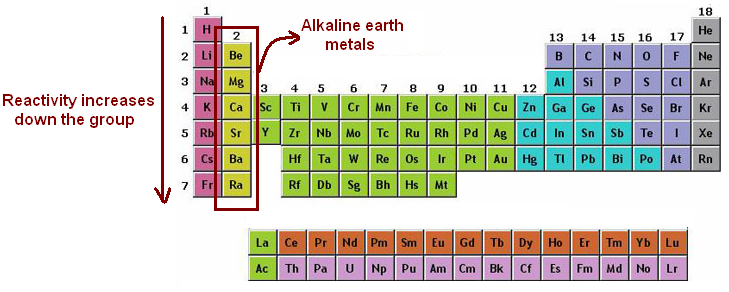
Sometimes cesium is also spelled "caesium.".Sodium and potassium both play an important role in biological life on Earth.Cesium clocks are considered the most accurate of all clocks. Cesium and rubidium are used to make atomic clocks.Because they are so reactive with air and water, they are generally stored in oil.The most abundant of the alkali metals on Earth is sodium. They have a low density when compared to other metals.They are malleable, ductile, and good conductors of electricity and heat.Some of them will even explode when they come into contact with water. They react when coming into contact with water. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
